INSIDE THE CAPSULE
Our History

2007
The CLOVER project was established in the year 2007, with the creation of the company and initial development of its product portfolio (comprising different pharmaceutical forms). Business activity was started in the Portuguese market.

2008
The company was established under the name “HC CLOVER” starting its operations in the Spanish market.

2009
The first soft gelatin capsule products were launched. The development and promotion of these products was started.

2010
The production plant in Arganda del Rey was built, and the first soft gelatin capsule production line was started. That same year, HC Clover received authorization for the manufacture of food supplements.

2011
In 2011, the company entered the European market, after operating in Portugal, Spain and France for a few years. The brand name “Aplicaps by Clover” was created that same year, and the second soft gelatin capsule production line was opened.

2012
In 2012, the company received authorization for the manufacture of medicinal products for human use, with the GMP certification. Expansion continued, and the company entered the Latin American market.

2013
Because of the growth of its turnover, a third soft gelatin capsule production line was created.
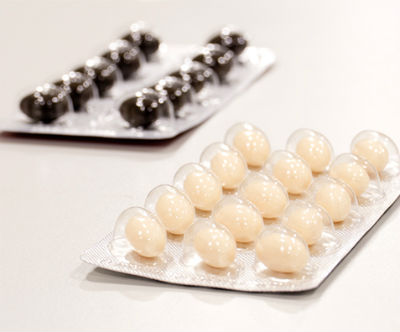
2014
In 2014, the company opened a fourth manufacturing line and a blister production line, making it possible to supply clients with completely finished products. Expansion continued into Europe and Latin America. The company was certified as a manufacturer of veterinary products.

2015
In 2015, the company created the gelatin processing plant and the pilot plant, which guarantee the viability of new products and the end quality of its products.
The fifth “Aplicaps by Clover” production line was opened.
"Aplicaps by Clover" was authorized as a manufacturer of ecological products.
The company started to expand into the Middle East and Eastern Europe.
Our Values
Our Team

CEO
Jose Luis Martín Guinea

R&D Manager
Blanca Rodríguez

Sales Manager
Iago Caamaño

Business Development Manager
Celia Secades

Supply Chain and
finance Manager
María José Cascajo

Qualified Person
José Manuel Albert
Discover the quality of our work
Spain
C/Alicante nº 8-10
Arganda del Rey, 28500, Madrid- +34 91 871 94 62
- +34 91 870 62 08
Portugal
Av José Malhoa Nº2, Esc.1.1
Edifício Malhoa Plaza, Lisboa- +35 210 939 458
United States
771 Shotgun Rd Sunrise, FL 33326
Contact: Angie Huffington
Brand Development- 786 518 2903
- brandevelop@ingpharmaceutical.com


